Magnetic resonance molecular imaging of neuroinflammation in mouse models of Alzheimer's and Parkinson's diseases by precision targeting of CSF1-R
Microglia-mediated neuroinflammation plays a pivotal role in the initiation and…
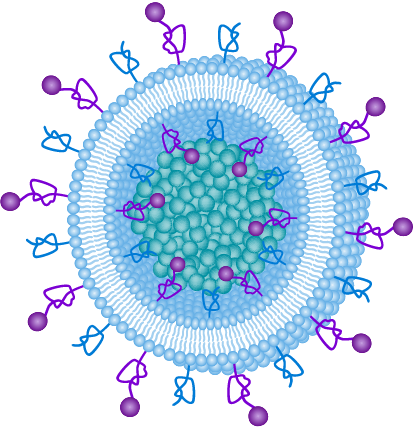
Engineered multifunctional nanoparticles for enhanced radiation therapy: three-in-one approach for cancer treatment
Abstract Clinical cancer treatment modalities include radiation as one of…
Flavocure Biotech, Inc. Announces a New Appointment to the Advisory Board
Baltimore, Maryland–(Newsfile Corp. – June 7, 2021) – The Board…
Former AstraZeneca Medical Director Joins Flavocure Biotech as Chief Medical Officer
BALTIMORE, MARYLAND, August 27, 2020 – For Immediate Release – Flavocure…
Flavonoid Derivative of Cannabis Demonstrates Therapeutic Potential in Preclinical Models of Metastatic Pancreatic Cancer
Abstract Pancreatic cancer is particularly refractory to modern therapies, with…